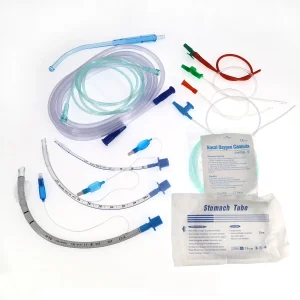
Ett Fixer Endotracheal Tubes Anesthesia Circuit Tubes Breathing OEM Single Use


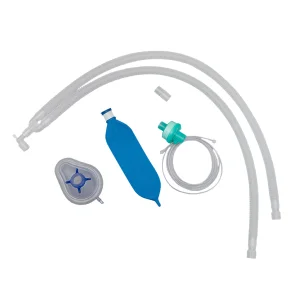
Comfortable Non-Toxic Materials Limb Breathing Ventilator Circuit Disposable Breathing Circuit

Safe Use Flexible Tube Single Pack Urinary Catheter for Urine Drainage
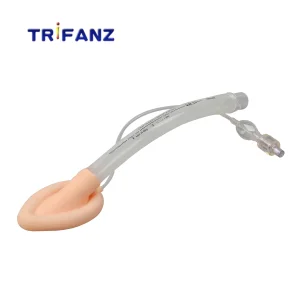

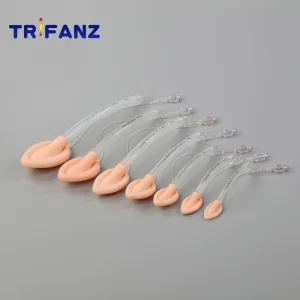
Disposable Surgical Silicone Laryngeal Mask Airway for Neonate Adult
The global dental materials market is undergoing a seismic shift, driven by an aging population, rising aesthetic awareness, and technological breakthroughs in regenerative medicine. As a premier OEM/ODM Dental Materials Supplier & Exporter, we recognize that the industry is no longer just about filling cavities; it is about systemic health integration. Today's commercial dental landscape demands materials that are not only biocompatible but also optimized for CAD/CAM (Computer-Aided Design and Manufacturing) workflows.
From Europe to Southeast Asia, dental professionals are seeking high-grade anesthesia circuits and oral surgical consumables that minimize cross-infection risks. The demand for restorative materials, impression compounds, and surgical masks has surged, particularly those that incorporate antimicrobial properties and enhanced durability. Global industrial stakeholders are prioritizing vertical integration in their supply chains, seeking partners who can offer both raw material excellence and finished product precision.
One of the most significant trends in the dental materials sector is the move toward "Smart Materials." These are substances designed to respond to pH changes or release fluoride and therapeutic ions over time. Furthermore, the integration of 3D printing in dental labs has revolutionized the production of crowns, bridges, and orthodontic aligners. This shift requires suppliers to be agile, offering OEM services that can customize silicone-based masks or PVC laryngeal airways to meet specific anatomical requirements for maxillofacial surgeries.
Eco-friendly manufacturing is also taking center stage. As a responsible exporter, we are seeing a massive increase in procurement requests for non-toxic, latex-free, and recyclable medical components. The dental industry is increasingly focused on reducing its environmental footprint while maintaining the highest levels of sterility and patient safety.
Our facility in Ningbo utilizes AI-driven quality monitoring systems to ensure every endotracheal tube and anesthesia mask meets micron-level tolerances. We bridge the gap between traditional craftsmanship and modern robotics.

Procurement officers for large-scale dental hospital chains and international medical distributors are shifting their focus from "lowest price" to "highest reliability and compliance." In the post-pandemic era, supply chain resilience is paramount. Buyers now look for manufacturers who can handle complex OEM requests—such as multi-lumen endotracheal tubes or specialized urinary catheters—while maintaining strict adherence to FDA, CE, and ISO standards.
Our role as a Chinese manufacturing powerhouse allows us to mitigate supply chain disruptions. By centralizing production near the major logistics hub of Shanghai, we offer a strategic advantage in transit times and shipping costs. This is particularly crucial for time-sensitive dental materials that require climate-controlled storage and rapid deployment to clinical settings.
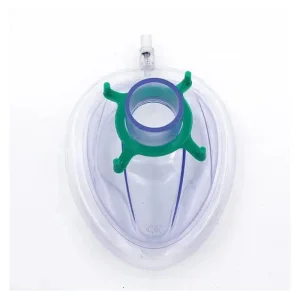
The application of our dental and surgical materials varies across global regions. In North America and Europe, there is a high demand for advanced anesthesia face masks used in outpatient dental sedation and pediatric dentistry. In developing markets, the focus is often on high-volume, sterile, disposable consumables like Foley catheters and urine bags for general hospital infrastructure. Our ODM capabilities allow us to tailor the physical properties of our products—such as tube flexibility or mask transparency—to meet the specific ergonomic preferences of local medical practitioners.
Ningbo Mainline Medical Co., Ltd. is a leading professional supplier of high-quality medical instruments and healthcare products in China. With over 10 years of experience in the medical field, our company has built a strong reputation for providing reliable and innovative solutions to meet the needs of healthcare professionals and patients worldwide.
Headquartered in Ningbo, Zhejiang Province, and strategically located near the port of Shanghai, we operate a state-of-the-art manufacturing facility where we produce a wide range of essential medical products, including syringes, infusion sets, gloves, and face masks. Our commitment to quality and innovation has made us a trusted partner for customers across Europe, Africa, Asia, and America.
To better serve our growing customer base, we have formed strong partnerships with over 5 strategic partners, each specializing in the production of complementary medical products such as urine bags, surgical gowns, medical tubes, wound dressings, syringes, diagnostic tools, and laboratory products. These collaborations ensure we can offer a comprehensive product portfolio that meets the diverse needs of healthcare facilities and distributors.
At Ningbo Mainline Medical, we understand that the foundation of healthcare lies in the quality and reliability of medical supplies. This is why we have established a robust quality control system, with a dedicated QC team monitoring every stage of production across all our product lines. We adhere to strict international standards to guarantee the highest level of quality and safety. Our company is fully certified with CE and ISO standards for most of our products, reflecting our commitment to delivering products that meet global quality and safety requirements.
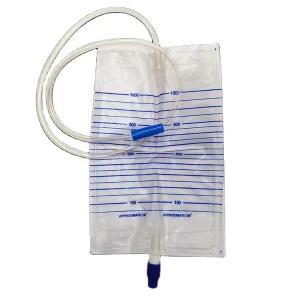
Medical Materials Sterile Medical Disposable Urine Bag Connector Adult Portable Drainage Urine Bag

Dual-Extruder Co-Extrusion Line for Color-Striped Multi-Lumen Endotracheal Tubes
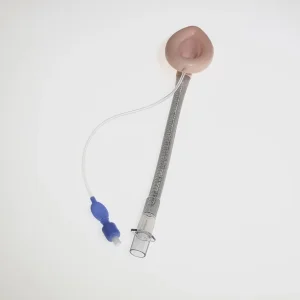
ISO CE Approved Medical Disposable Silicone Laryngeal Mask Airway Suitable for Children and Adults
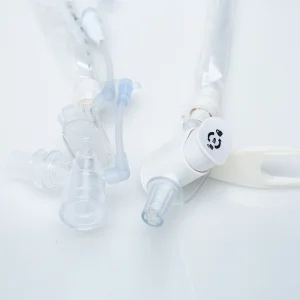

Catheter for Pyonephrosis Drainage Top-Quality Medical Instrument Ureteral Drainage Tube

Disposable Medical Urianry Foley Catheter for Women Female Male Latex or Silicon