🔍 Product Description
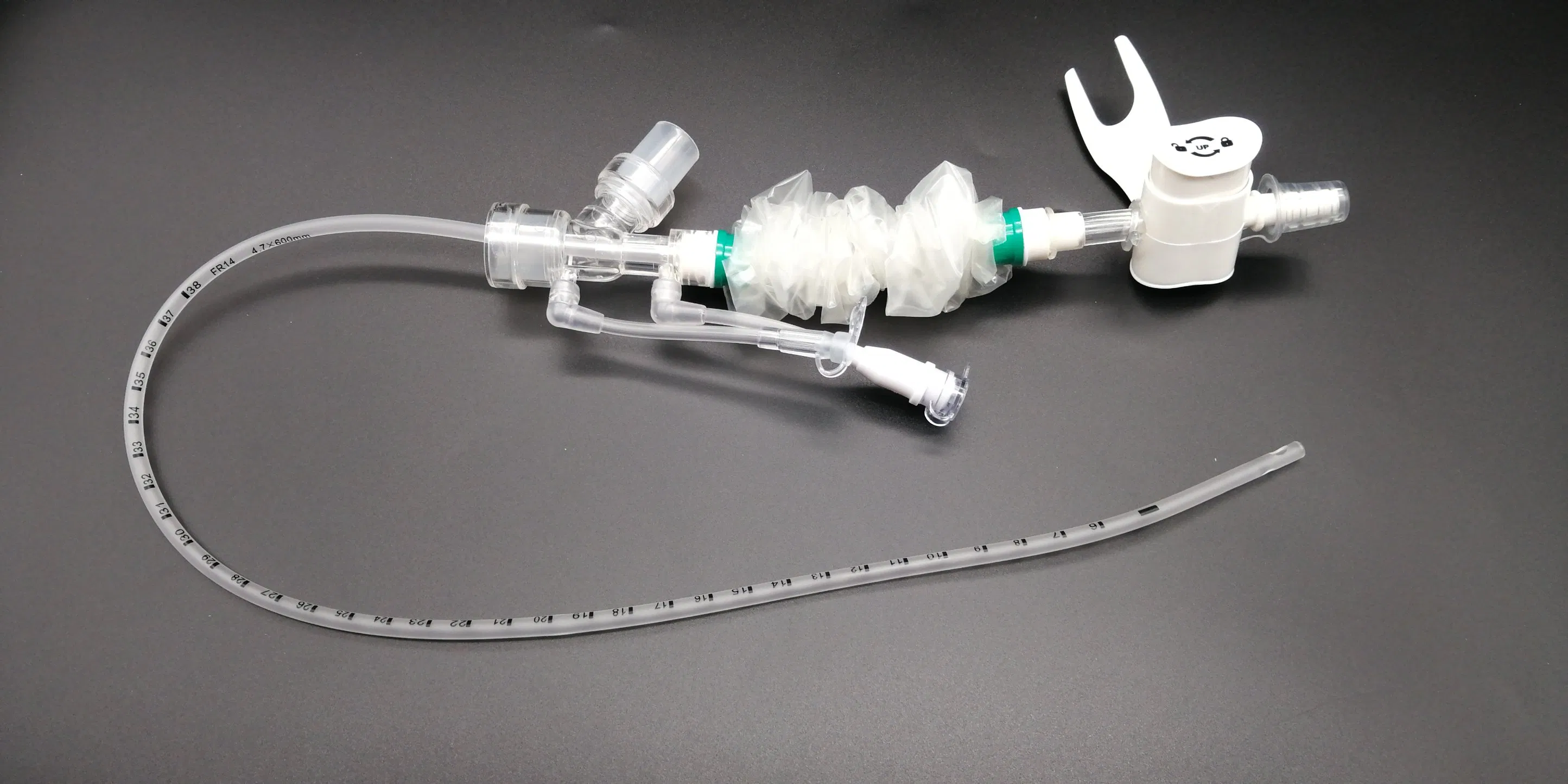
Surgical Disposable PVC Medical Suction Tube Catheter with 72 Hour Mark
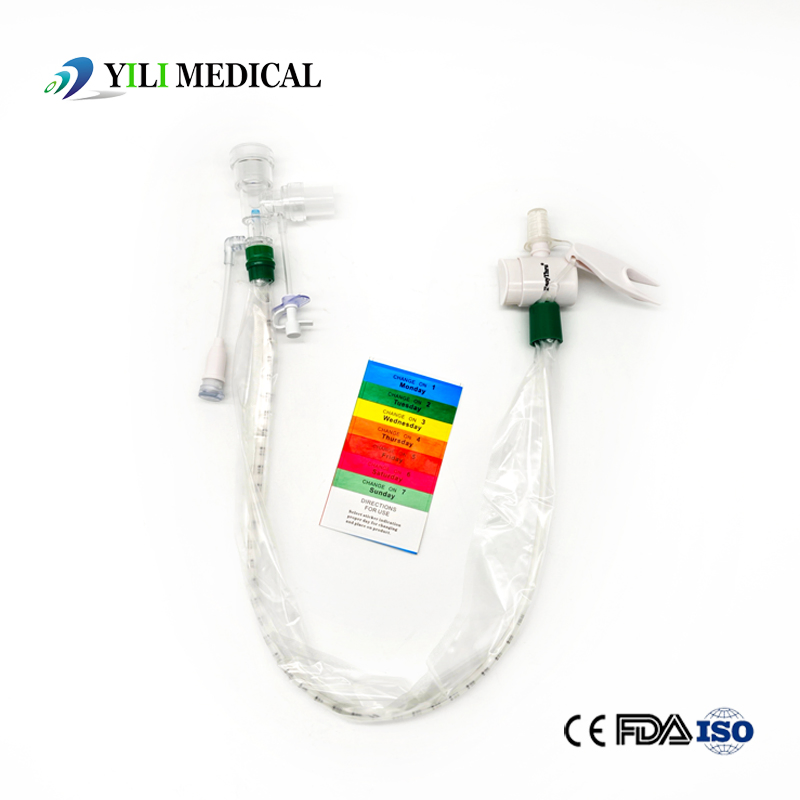
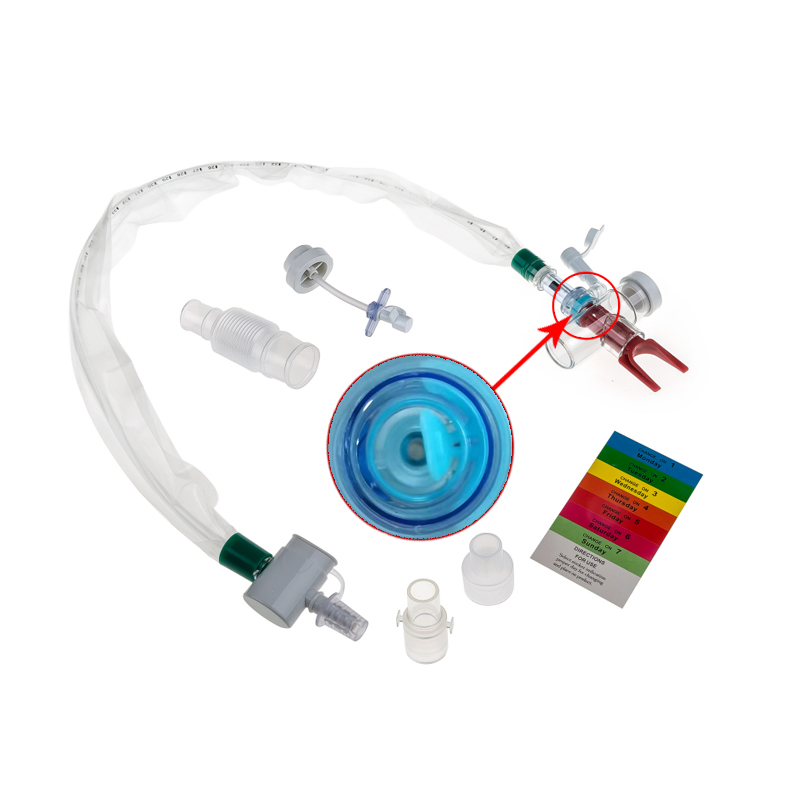
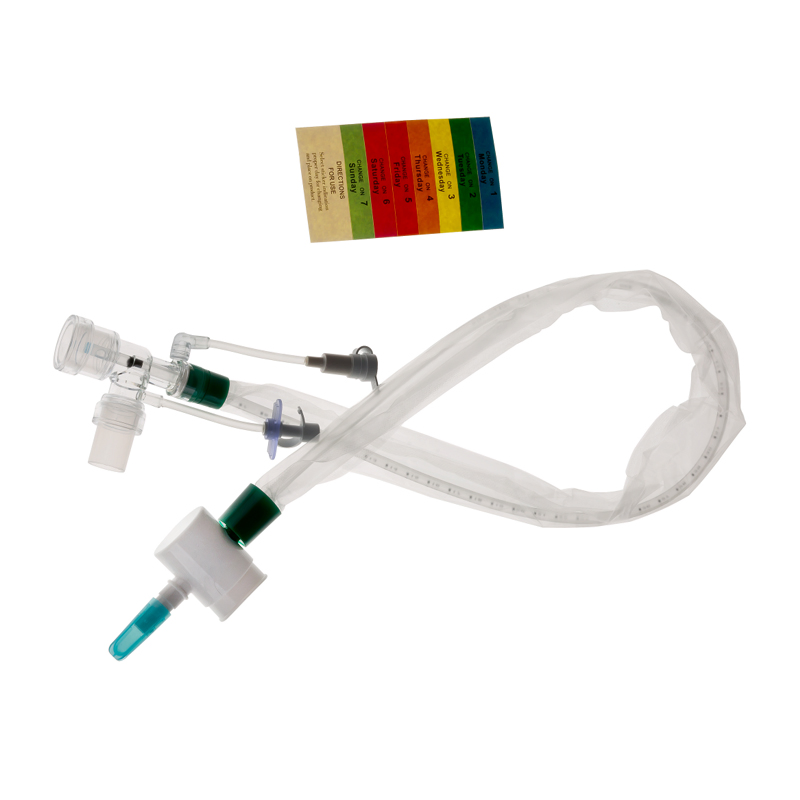
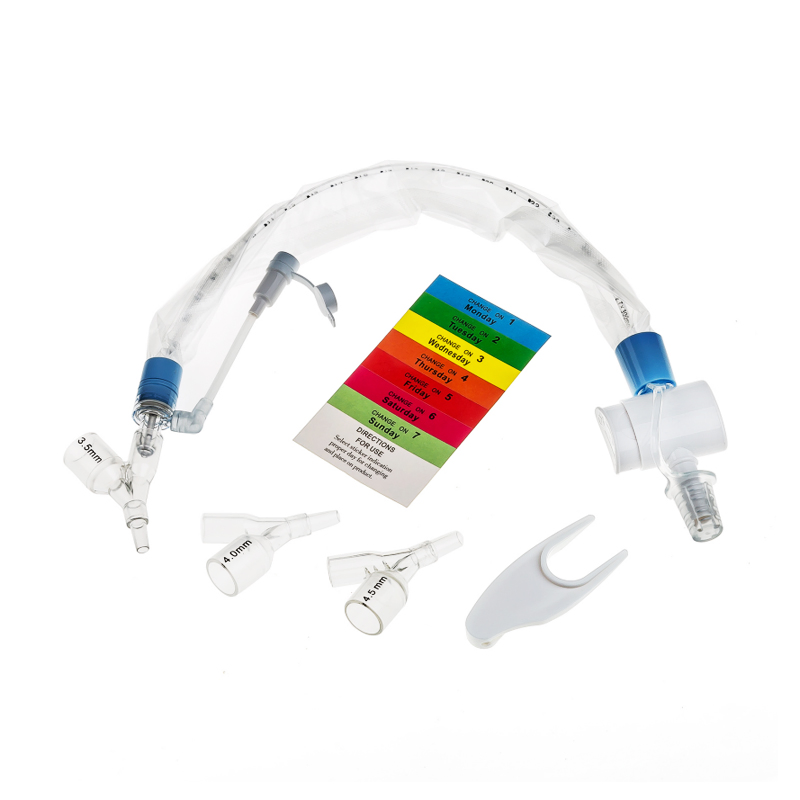
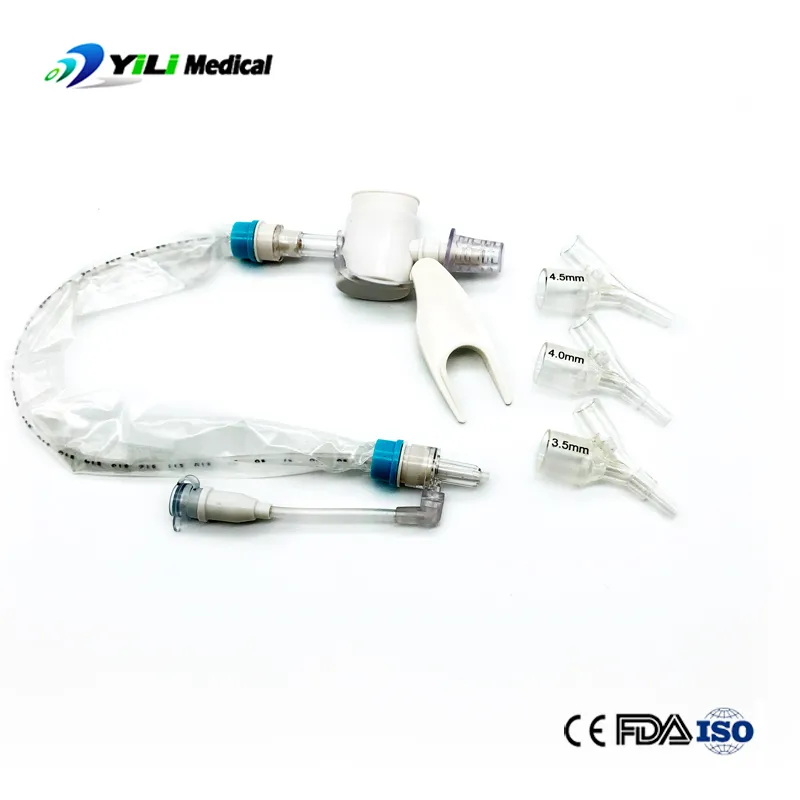
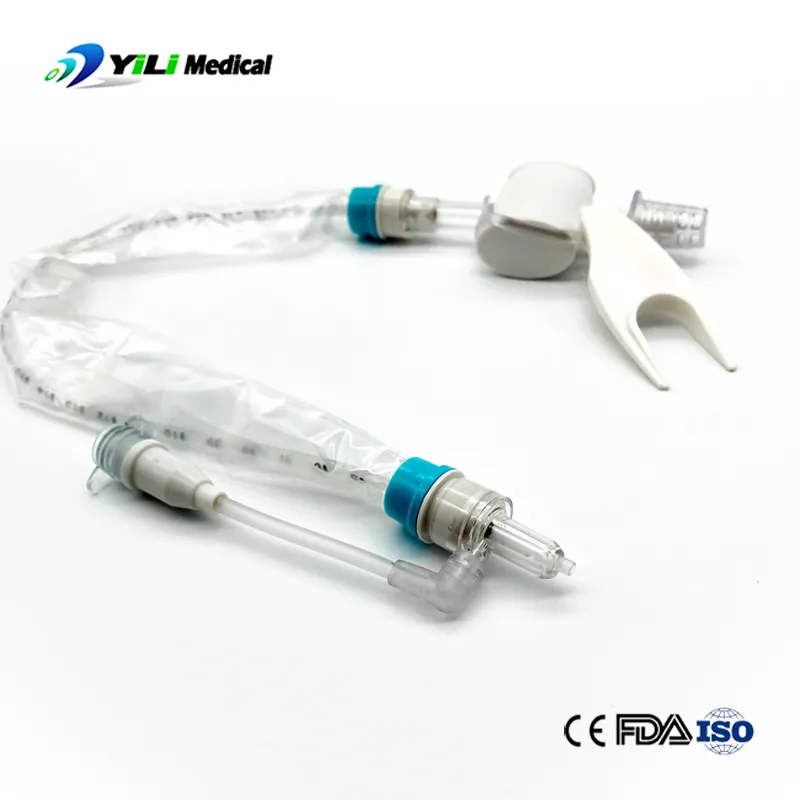
What is Closed Suction System?
The Closed Suction System is intended for removal of secretion in the lower respiratory track in patients receiving prolonged mechanical ventilation. The filter protected system is connected to the artificial airway of the patient (Endotracheal or Tracheostomy tube) and to the ventilation circuit. It is used over a period of 24 hours or 72 hours, depending on the patient requirement.
Product size range: Fr6, Fr8, Fr10, Fr12, Fr14, Fr16. Y Connector Type available for 6, 7, 8, French sizes closed suction catheter.
Packaging: Blister individually, 100 pcs/ carton
| Application |
Personal Care |
| Properties |
Medical Materials & Accessories |
| After-sale Service |
Online technical support |
💡 Why use closed suction system?
- Continuous Ventilation: Traditional suction requires cutting off the airway. The closed system allows for suction without interrupting the patient's oxygen supply.
- Cost-Effective: Over a 48-hour period, the average cost is comparable to traditional catheters given the frequency of suctioning required in ICU settings.
- Infection Control: Reduces cross-infection risks. The catheter is covered by a sleeve, preventing secretions from splashing when pulled out, protecting both patients and medical staff.
🏥 Product Scope
Suitable for respiratory system diseases, general anesthesia, surgical treatment, and emergency rescue involving endotracheal or tracheostomy tubes. It effectively extracts secretions during artificial ventilator support.
🏭 Professional Manufacturing
With over 9 years of expertise, the manufacturing process covers breathing anesthesia products, urinary, and injection infusion supplies. Family-owned facilities manage different product lines to offer a comprehensive one-stop service.
Product Qualification:
- Operated under ISO 13485 quality control system.
- All products are CE certificated.
- Produced in Cleanroom environments following strict SOPs for traceability.
- Finished goods testing reports provided before shipment.
📦 Packaging & Transportation
❓ FAQ
1. Are you a trading company?
No. We are a manufacturer with 9 years of experience in Disposable Medical products, serving customers in over 88 countries worldwide.
2. How about the lead time?
About 15-30 working days after receiving payment and confirming all artworks. The exact time depends on the order quantity and packaging requirements.
3. Can our private logo / label be printed on the packaging?
Yes, your private logo or label can be printed upon legal authorization. We have provided OEM services for many years.
4. How can I get some samples?
We provide some free samples; however, postage is covered by the requester. These charges can be deducted from the final order payment. Alternatively, you can provide a carrier account (DHL, UPS, etc.).
5. What is the best price you can offer?
We work hard to provide competitive pricing while maintaining high quality to satisfy market demands. Please send your specific inquiry for a detailed quote.
6. Why choose us?
We have passed FDA, CE, ISO, FSC, and GMP certifications, ensuring the best service and high quality at competitive prices.