■ Basic Information
Model NO.
1.0#/1.5#/2.0#/2.5#/3.0#/4.0#/5.0#
Sterilization
Ethylene Oxide Sterilization
Quality Guarantee
Three Years
Name
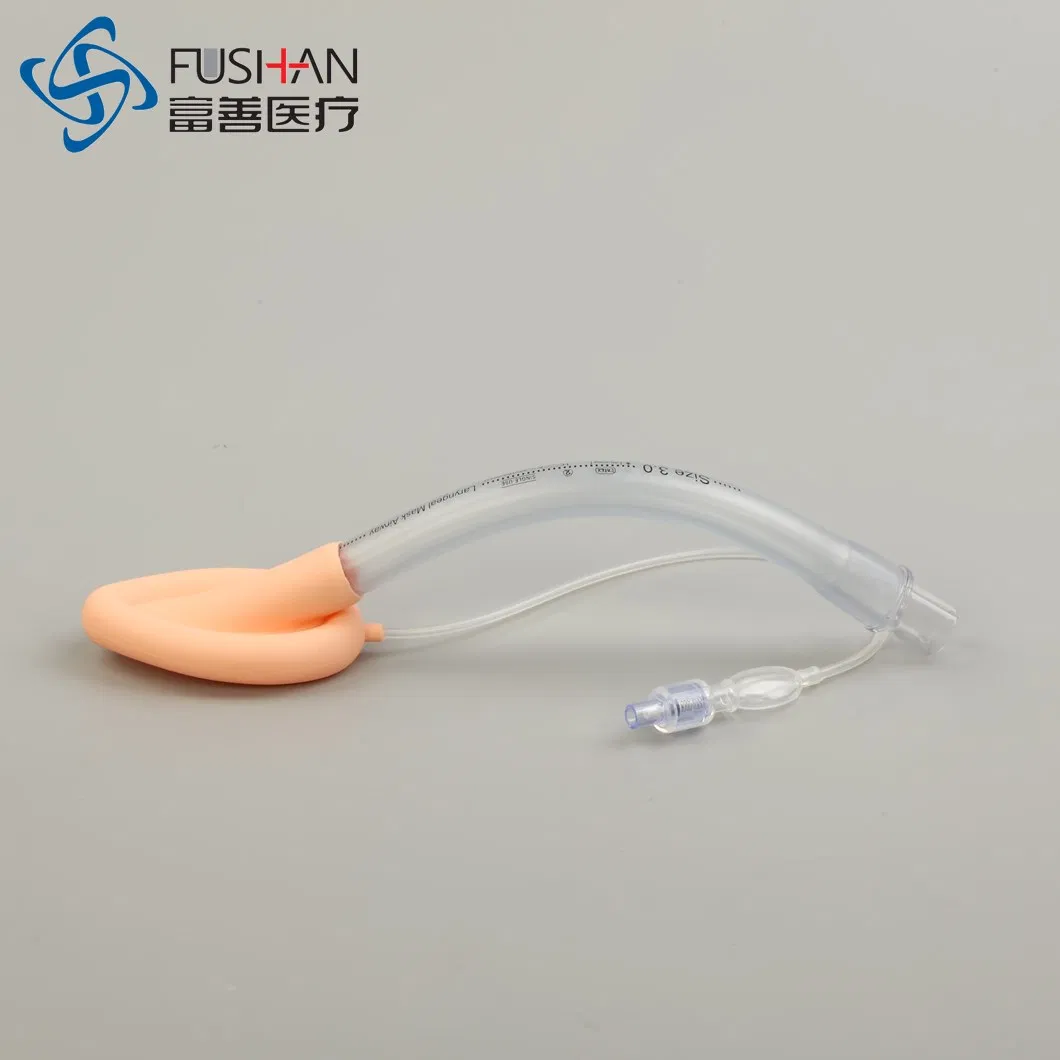
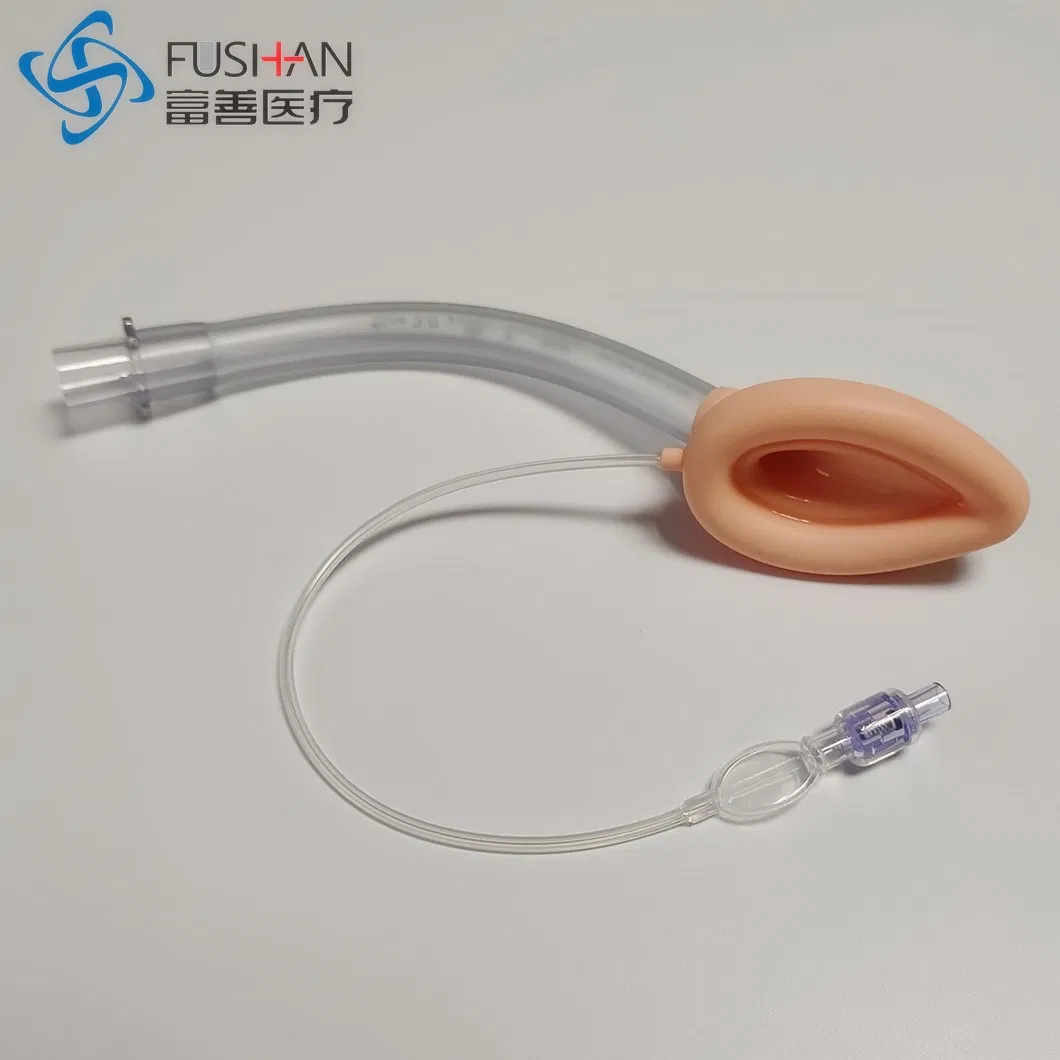
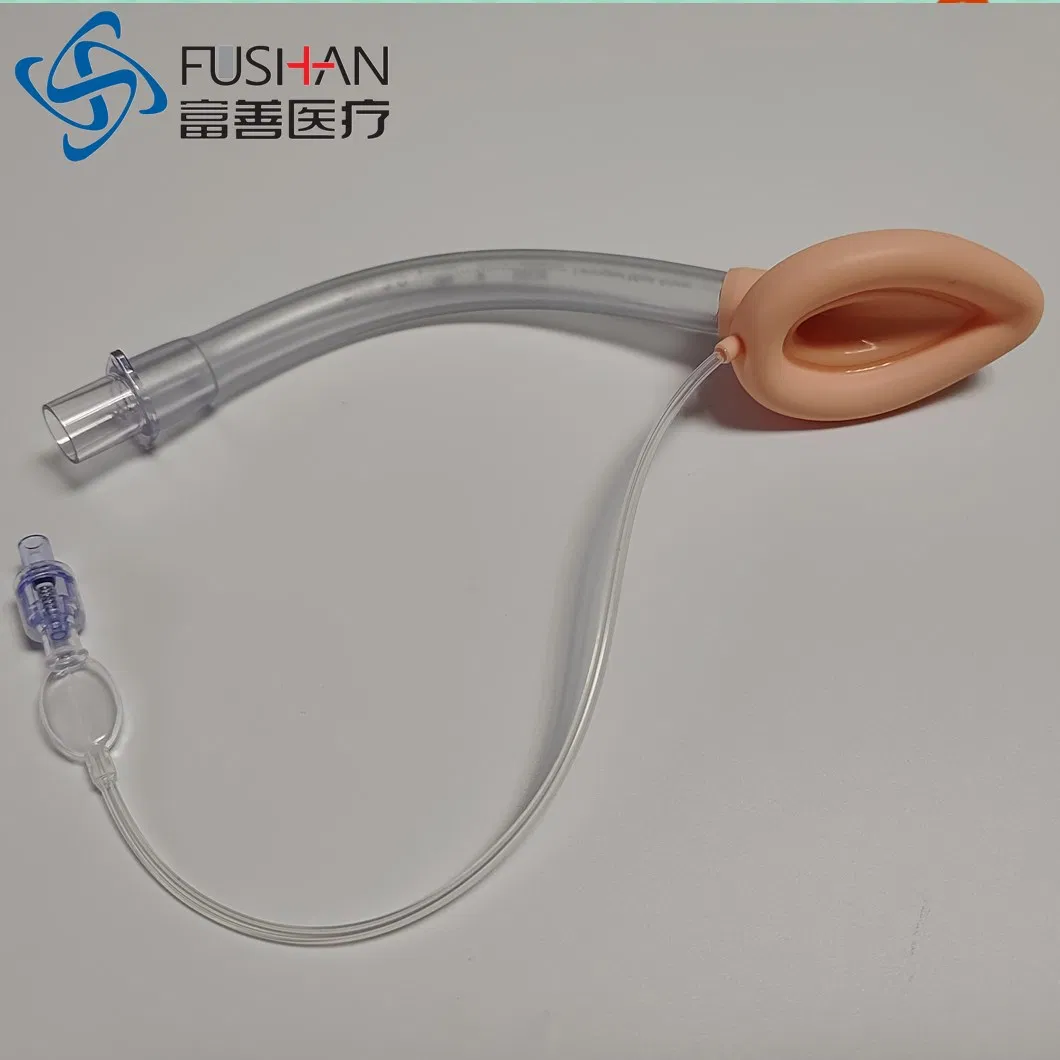
PVC-Silicone Combined Laryngeal Mask Airway
Characteristic
Cuff Softer Than PVC
■ Product Description
PVC-Silicone Laryngeal Mask Airway
The laryngeal mask is used for general anesthesia surgery, ensuring a stable breathing passage for patients. It seals the laryngeal cavity and prevents secretions from entering, which protects respiratory function during procedures. Unlike traditional endotracheal intubation, this laryngeal mask avoids direct tracheal insertion, reducing the risk of laryngeal injury, arytenoid joint dislocation, and mucosal damage. It is an ideal solution for general anesthesia except in cases involving upper respiratory tract surgery.
Key Advantages:
✓ Tube made of medical-grade PVC; cuff made of medical-grade silicone.
✓ Combines PVC's structural integrity with silicone's superior comfort.
✓ Simple operation: no laryngoscope required for glottis exposure.
✓ Reduced post-operative complications like sore throat or edema.
Contraindications:
• Patients with high intra-abdominal pressure or risk of vomiting/aspiration.
• Throat infections or pathological changes.
• Surgery requiring positive pressure ventilation above 25cmH20.
• Abnormalities such as enlarged tonsils or limited mouth opening.
■ Size Specifications
| Item NO. |
Size(Fr) |
Max Cuff Volume(ml) |
Patient Weight(KG) |
| LM1004P1 | 1.0 | 4 | 0-5 |
| LM1507P1 | 1.5 | 7 | 5-10 |
| LM2010P1 | 2.0 | 10 | 10-20 |
| LM2514P1 | 2.5 | 14 | 20-30 |
| LM3020P1 | 3.0 | 20 | 30-50 |
| LM4030P1 | 4.0 | 30 | 50-70 |
| LM5040P1 | 5.0 | 40 | 70-100 |
■ Production Capability
The manufacturing facility covers an area of 13,000 sqms, featuring class 10,000 and class 100,000 clean workshops designed according to GMP standards. Equipped with precise manufacture and inspection facilities, we provide high-quality medical devices made of silicone, polyurethane (PU), and polyvinyl chloride (PVC). Our professional labs are dedicated to sterile, physicochemical, and electrical tests, supported by CE-verified ETO sterilization chambers.
■ Frequently Asked Questions
Q1: What kind of certificates do you have?
A: We are a professional manufacturer with CE, ISO, SGS, and other relevant certificates required for international markets.
Q2: Can you provide samples?
A: Yes, we can provide sample productions and delivery services for quality evaluation. Please contact our team for details.
Q3: How do you ensure product quality?
A: We implement strict quality control including material inspection, in-process inspection, and a final full inspection before shipment.
Q4: Do you offer OEM/ODM services?
A: Yes, we provide full OEM and ODM services, including customized packaging and logo printing.
Q5: What is the lead time for mass production?
A: The lead time typically ranges between 30 to 45 days, depending on the order quantity and seasonal demand.
Q6: What is the standard packaging?
A: Our standard packaging is an individual sterile blank paper-plastic bag. Custom OEM packaging is also available.