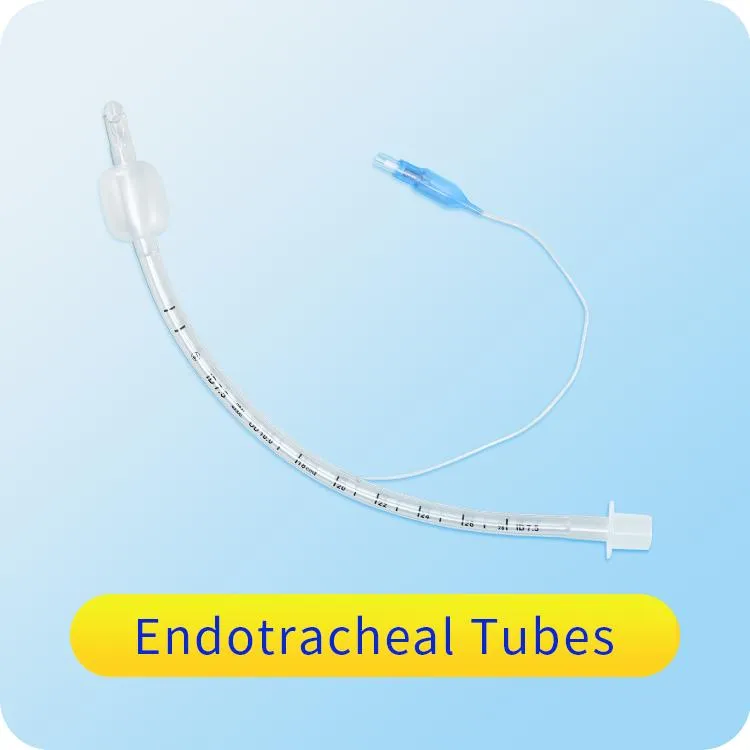
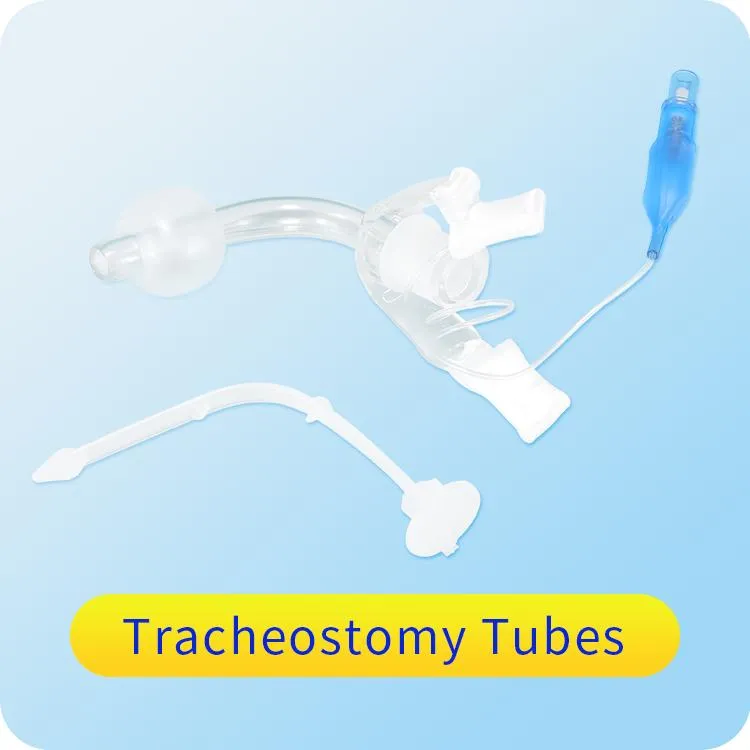
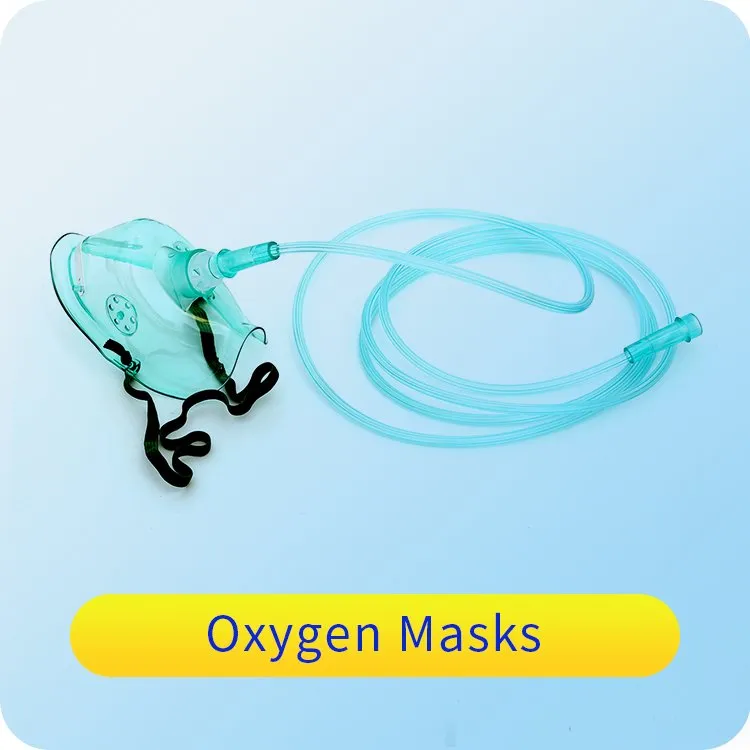
1 / 5










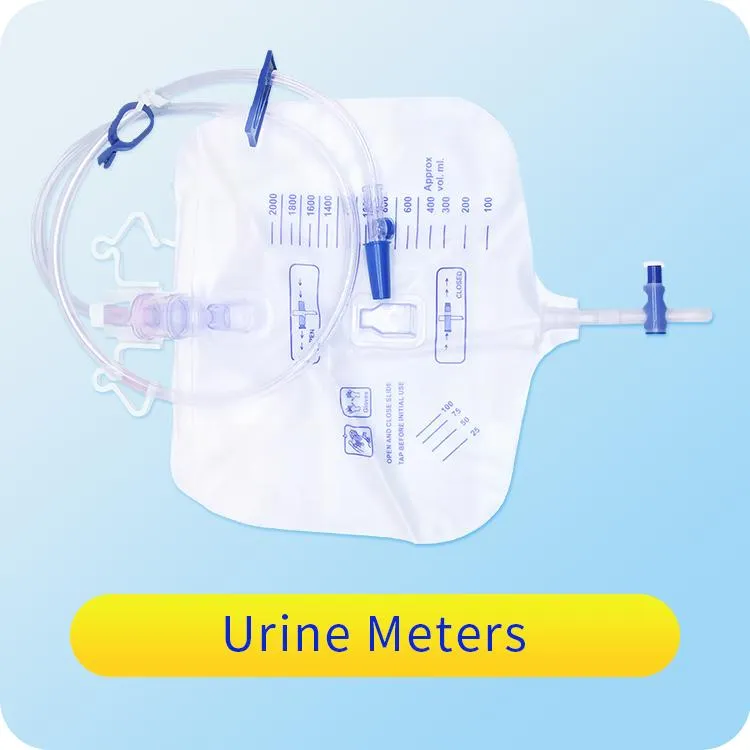
The Urine Bag is manufactured from high-quality medical polymeric materials (PVC). The complete set includes a urine collection bag, inlet tubing, outlet tubing, restraint, connector, protective cap, drainage tap, and fastening device.
| Ref. NO. | Capacity Size |
|---|---|
| KR-UBT01000 | 1000ml |
| KR-UBT02000 | 2000ml |





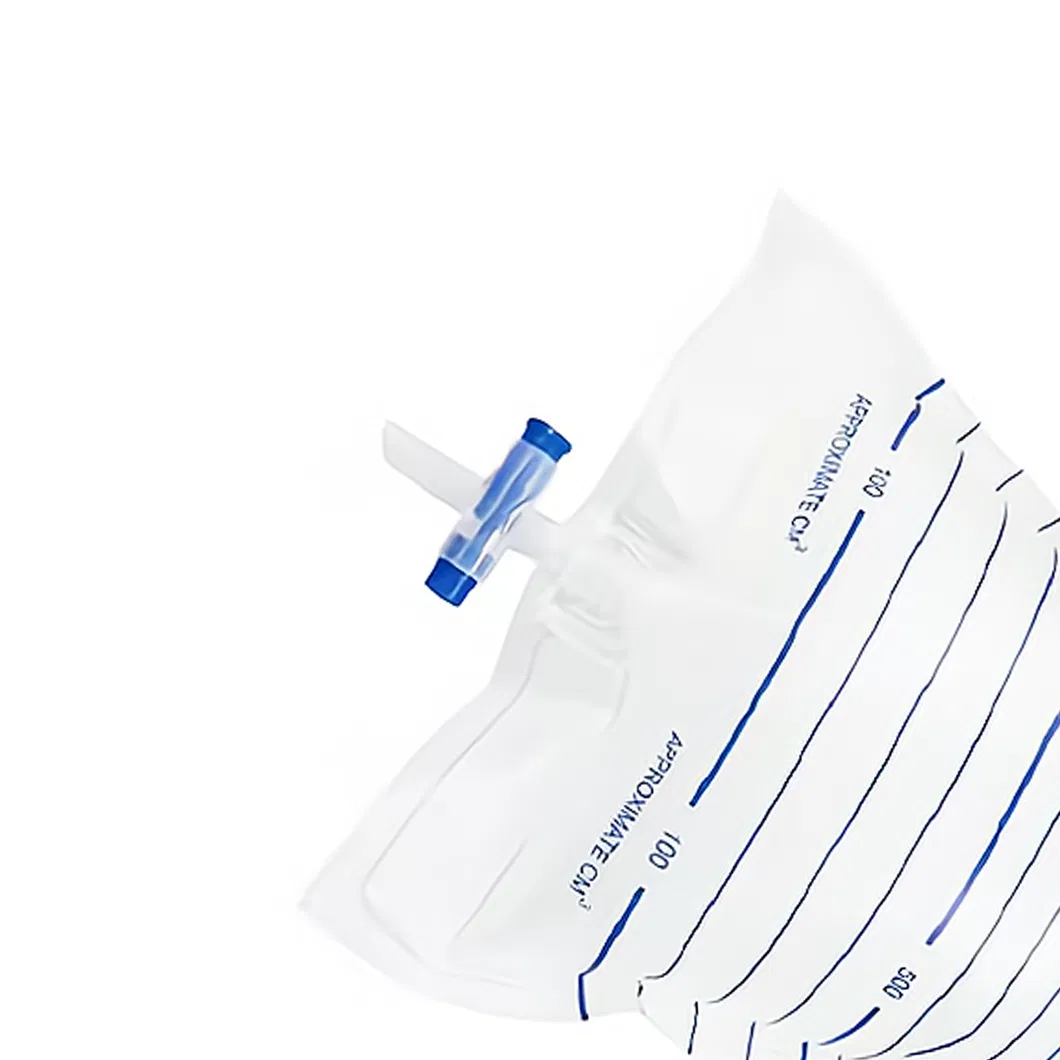
Designed for the drainage and collection of body fluids from patients in clinical environments.