











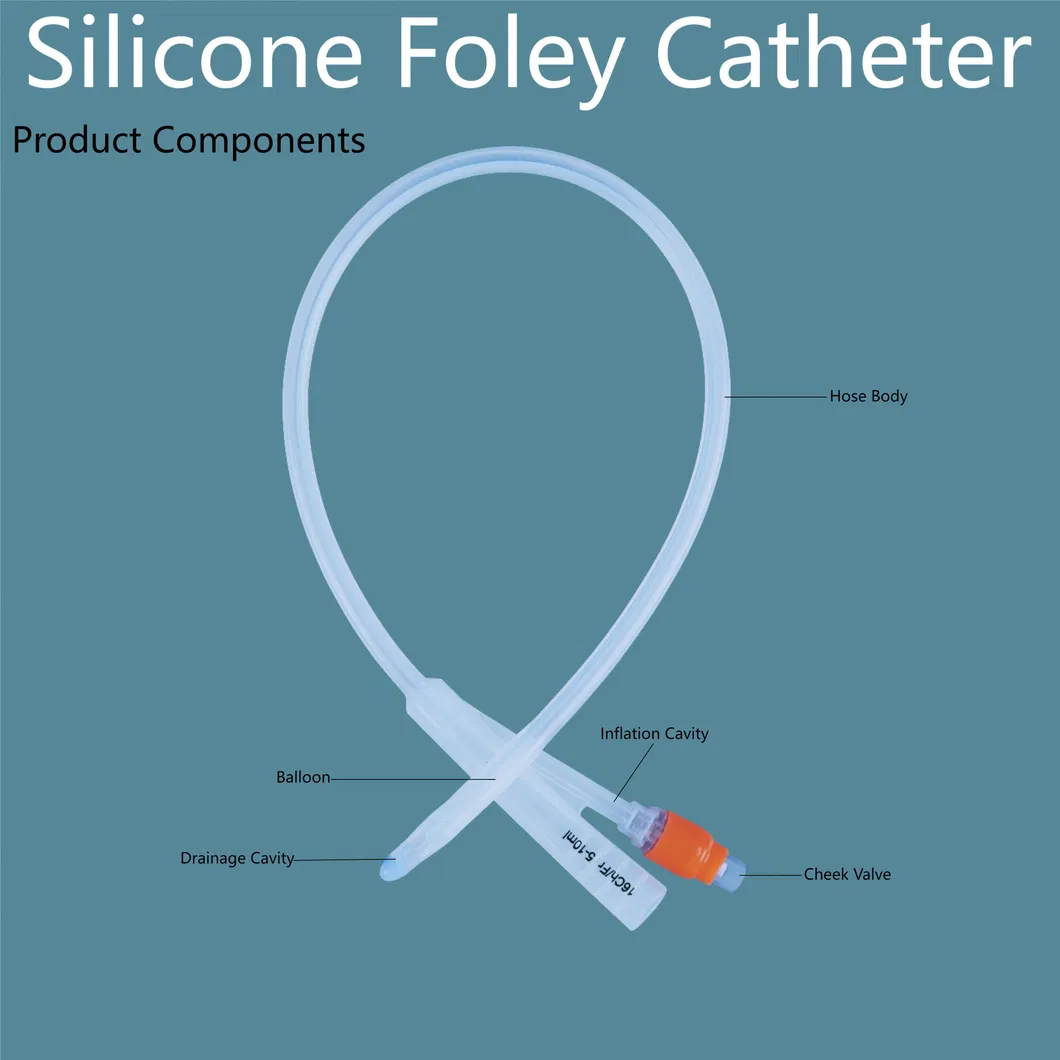
Silicone Foley Catheter is used in departments of urology, internal medicine, surgery, obstetrics, and gynecology for drainage of urine and medication. It is specifically designed for patients suffering from mobility difficulties or those who are bed-ridden.
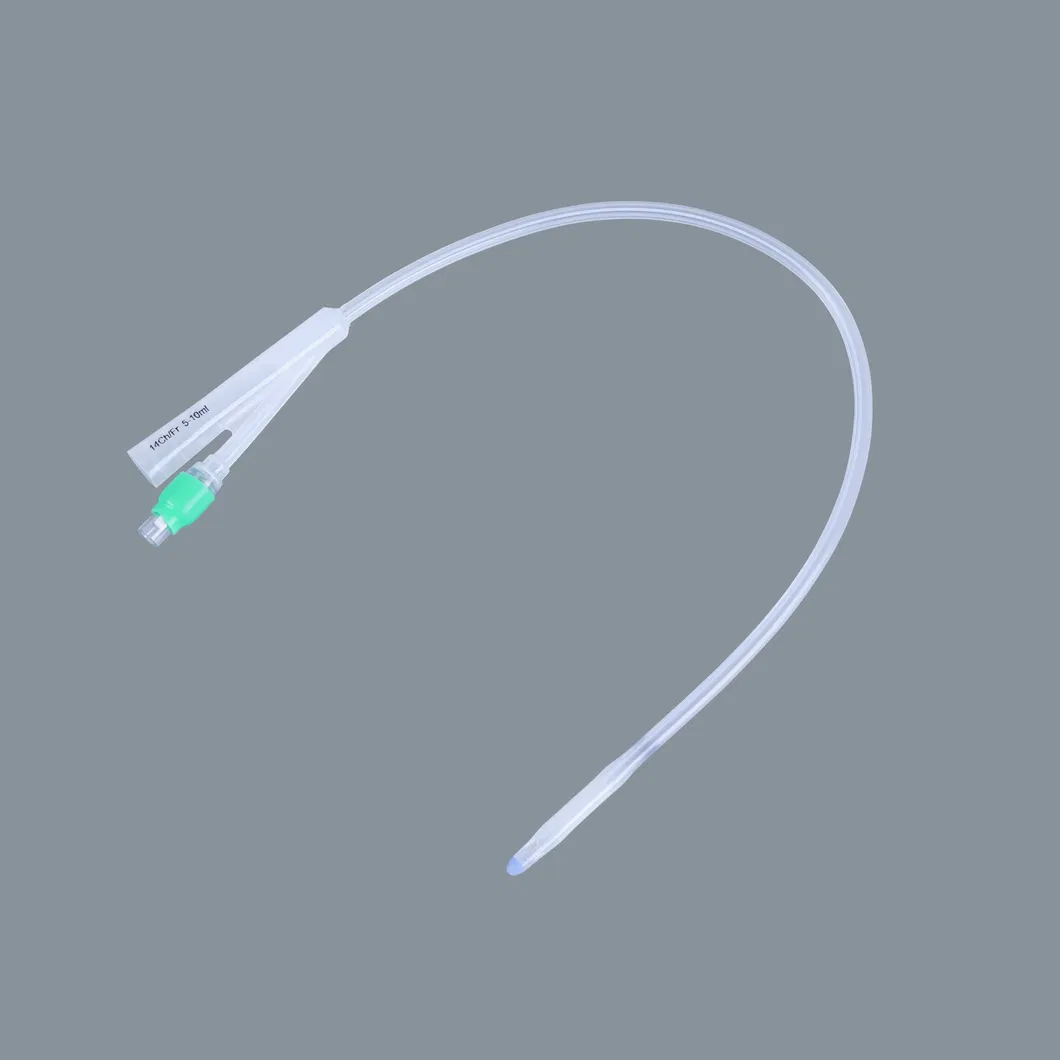

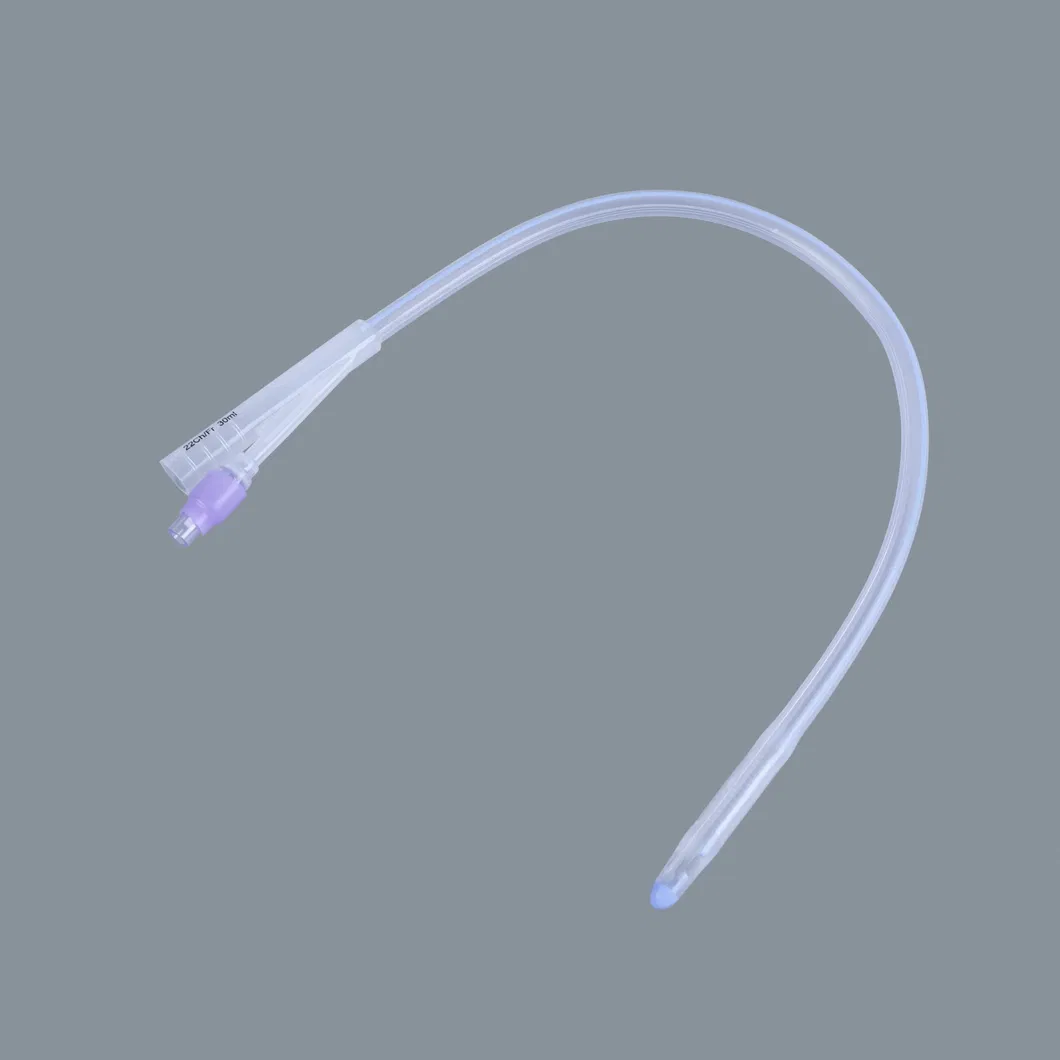
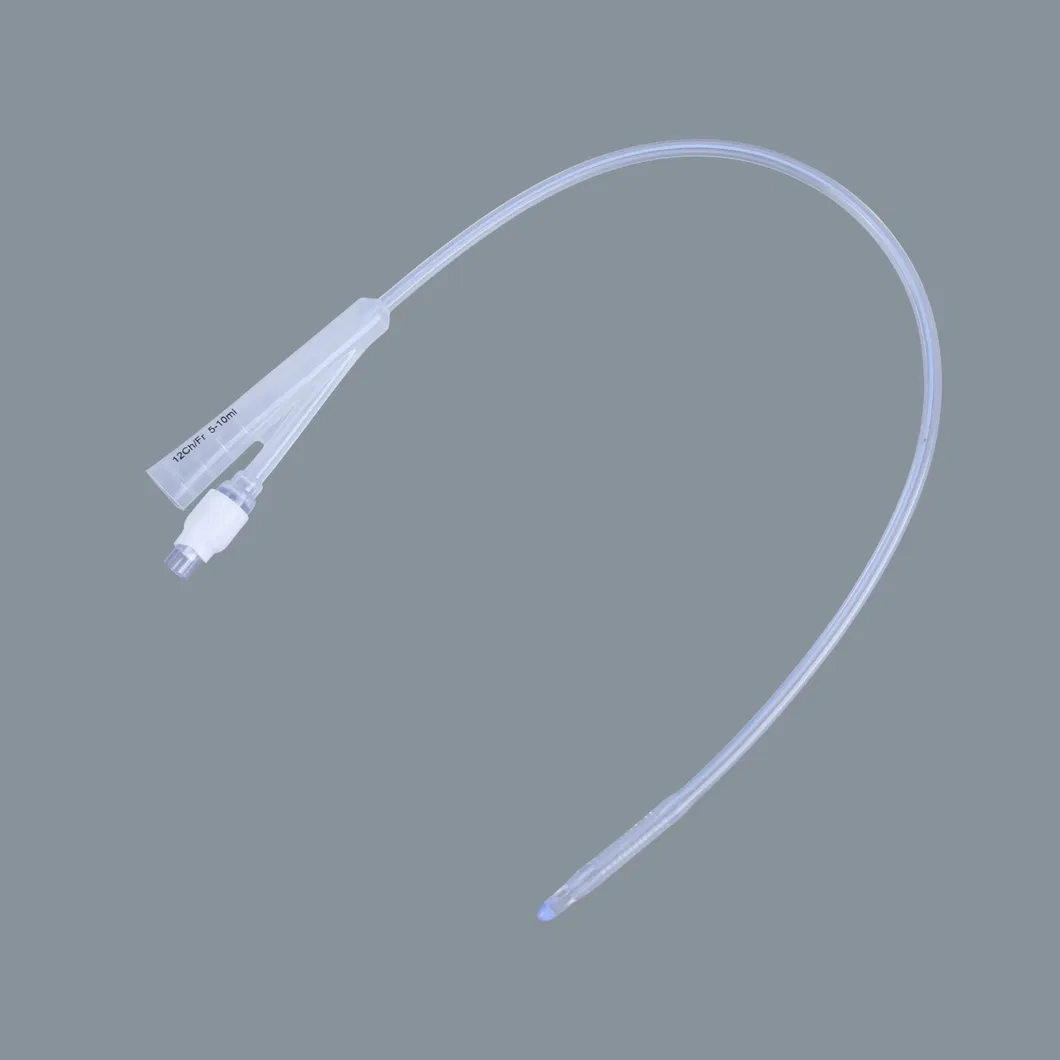
| Item | Specification | Packaging (pcs/ctn) | Weight (Net/Gross) | Volume (CBM) |
|---|---|---|---|---|
| Silicone Foley Catheter | 2-way (Fr6 - Fr26) 3-way (Fr12 - Fr24) |
500pcs | 9KGS / 11KGS | 0.098 CBM |
1. Remove catheter from sterile pouch using aseptic technique. Inspect for mechanical damage. Only sterile water should be used to fill the balloon.
Insertion:2. Inform patient about the procedure and possible discomfort.
3. Verify correct size and check balloon integrity.
4. Wash hands thoroughly and disinfect the perineum.
5. Use appropriate lubricant and insert gently. Once urine flows, insert 5cm-6cm further.
6. Inflate the balloon with sterile water and pull gently until resistance is felt.